Alfred Werner
Alfred Werner | |
|---|---|
 Werner circa 1915 | |
| Born | 12 December 1866 Mulhouse, Haut-Rhin, Alsace, France |
| Died | 15 November 1919(1919-11-15) (aged 52) Zürich, Switzerland |
| Nationality | Swiss (from 1895) French |
| Alma mater | University of Zurich ETH Zurich |
| Known for | Configuration of transition metal complexes |
| Spouse | Emma Werner[1] |
| Awards | Nobel Prize for Chemistry (1913) |
| Scientific career | |
| Fields | Inorganic chemistry |
| Institutions | University of Zurich |
| Doctoral advisor | Arthur Rudolf Hantzsch, Marcellin Berthelot[citation needed] |
Alfred Werner (12 December 1866 – 15 November 1919) was a Swiss chemist who was a student at ETH Zurich and a professor at the University of Zurich. He won the Nobel Prize in Chemistry in 1913 for proposing the octahedral configuration of transition metal complexes. Werner developed the basis for modern coordination chemistry. He was the first inorganic chemist to win the Nobel Prize, and the only one prior to 1973.[2]
Biography
Werner was born in 1866 in Mulhouse, Alsace (which was then part of France, but which was annexed by Germany in 1871). He was raised as Roman Catholic.[3] He was the fourth and last child of Jean-Adam Werner, a foundry worker, and his second wife, Salomé Jeanette Werner, who originated from a wealthy family.[3] He went to Switzerland to study chemistry at the Swiss Federal Institute (polytechnikum) in Zurich. Still, since this institute was not empowered to grant doctorates until 1909, Werner received a doctorate formally from the University of Zürich in 1890.[3] After postdoctoral study in Paris, he returned to the Swiss Federal Institute to teach (1892). In 1893 he moved to the University of Zurich, where he became a professor in 1895. In 1894 he became a Swiss citizen.[3]
In his last year, he suffered from a general, progressive, degenerative arteriosclerosis, especially of the brain, aggravated by years of excessive drinking and overwork. He died in a psychiatric hospital in Zürich.[3]
Werner died on 15 November 1919 of arteriosclerosis in Zürich at the age of 52.
Research
Coordination chemistry
In 1893, Werner was the first to propose correct structures for coordination compounds containing complex ions, in which a central transition metal atom is surrounded by neutral or anionic ligands.
For example, it was known that cobalt forms a "complex" hexamine cobalt (III) chloride, with formula CoCl3•6NH3, but the nature of the association indicated by the dot was mysterious. Werner proposed the structure [Co(NH3)6]Cl3, with the Co3+ ion surrounded by six NH3 at the vertices of an octahedron. The three Cl− are dissociated as free ions, which Werner confirmed by measuring the conductivity of the compound in an aqueous solution, and also by chloride anion analysis using precipitation with silver nitrate. Later, magnetic susceptibility analysis was also used to confirm Werner's proposal for the chemical nature of CoCl3•6NH3.

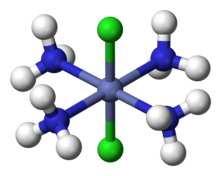
For complexes with more than one type of ligand, Werner succeeded in explaining the number of isomers observed. For example, he explained the existence of two tetramine isomers, "Co(NH3)4Cl3", one green and one purple. Werner proposed that these are two geometric isomers of formula [Co(NH3)4Cl2]Cl, with one Cl− ion dissociated as confirmed by conductivity measurements. The Co atom is surrounded by four NH3 and two Cl ligands at the vertices of an octahedron. The green isomer is "trans" with the two Cl ligands at opposite vertices, and the purple is "cis" with the two Cl at adjacent vertices.
Werner also prepared complexes with optical isomers, and in 1914 he reported the first synthetic chiral compound lacking carbon, known as hexol with formula [Co(Co(NH3)4(OH)2)3]Br6.
Nature of valence
Before Werner, chemists defined the valence of an element as the number of its bonds without distinguishing different types of bonds. However, in complexes such as [Co(NH3)6]Cl3 for example, Werner considered that the Co-Cl bonds correspond to a "primary" valence of 3 at long distance, while the Co-NH3 bonds which correspond to a "secondary" or weaker valence of 6 at shorter length. This secondary valence of 6 he referred to as the coordination number which he defined as the number of molecules (here of NH3) directly linked to the central metal atom. In other complexes, he found coordination numbers of 4 or 8.
On these views, and other similar views, in 1904 Richard Abegg formulated what is now known as Abegg's rule which states that the difference between the maximum positive and negative valence of an element is frequently eight. This rule was used later in 1916 when Gilbert N. Lewis formulated the "octet rule" in his cubical atom theory.
In modern terminology, Werner's primary valence corresponds to the oxidation state, and his secondary valence is called coordination number. The Co-Cl bonds (in the above example) are now classed as ionic, and each Co-N bond is a coordinate covalent bond between the Lewis acid Co3+ and the Lewis base NH3.
Works
- Lehrbuch der Stereochemie . Fischer, Jena 1904 Digital edition by the University and State Library Düsseldorf
References
- W. Gregory Jackson; Josephine A. McKeon; Silvia Cortez (2004). "Alfred Werner's Inorganic Counterparts of Racemic and Mesomeric Tartaric Acid: A Milestone Revisited". Inorg. Chem. 43 (20): 6249–6254. doi:10.1021/ic040042e. PMID 15446870.
- Kristin Bowman-James (2005). "Alfred Werner Revisited: The Coordination Chemistry of Anions". Acc. Chem. Res. 38 (8): 671–678. doi:10.1021/ar040071t. PMID 16104690.
- ^ "Alfred Werner - Nobel Prize for Chemistry 1913" (PDF). University of Zurich. Retrieved 9 December 2022.
He moved there with his wife, Emma Wilhelmine, née Giesker, whom he had married on 1 October 1894.
- ^ https://www.nobelprize.org/nobel_prizes/chemistry/laureates/1913/werner-bio.html Nobel Prize Retrieved 1 December 2012
- ^ a b c d e "Alfred Werner - Swiss chemist". britannica.com. Retrieved 14 April 2018.
External links
- Alfred Werner on Nobelprize.org
 including the Nobel Lecture, 11 December 1913 On the Constitution and Configuration of Higher-Order Compounds
including the Nobel Lecture, 11 December 1913 On the Constitution and Configuration of Higher-Order Compounds - The Nobel Prize in Chemistry 1913 - short article about his work on the linkage of atoms in molecules by which he has thrown new light on earlier investigations and opened up new fields of research, especially in inorganic chemistry.
- v
- t
- e
- 1901: Jacobus van 't Hoff
- 1902: Emil Fischer
- 1903: Svante Arrhenius
- 1904: William Ramsay
- 1905: Adolf von Baeyer
- 1906: Henri Moissan
- 1907: Eduard Buchner
- 1908: Ernest Rutherford
- 1909: Wilhelm Ostwald
- 1910: Otto Wallach
- 1911: Marie Curie
- 1912: Victor Grignard / Paul Sabatier
- 1913: Alfred Werner
- 1914: Theodore Richards
- 1915: Richard Willstätter
- 1916
- 1917
- 1918: Fritz Haber
- 1919
- 1920: Walther Nernst
- 1921: Frederick Soddy
- 1922: Francis Aston
- 1923: Fritz Pregl
- 1924
- 1925: Richard Zsigmondy

- 1926: Theodor Svedberg
- 1927: Heinrich Wieland
- 1928: Adolf Windaus
- 1929: Arthur Harden / Hans von Euler-Chelpin
- 1930: Hans Fischer
- 1931: Carl Bosch / Friedrich Bergius
- 1932: Irving Langmuir
- 1933
- 1934: Harold Urey
- 1935: Frédéric Joliot-Curie / Irène Joliot-Curie
- 1936: Peter Debye
- 1937: Norman Haworth / Paul Karrer
- 1938: Richard Kuhn
- 1939: Adolf Butenandt / Leopold Ružička
- 1940
- 1941
- 1942
- 1943: George de Hevesy
- 1944: Otto Hahn
- 1945: Artturi Virtanen
- 1946: James B. Sumner / John Northrop / Wendell Meredith Stanley
- 1947: Robert Robinson
- 1948: Arne Tiselius
- 1949: William Giauque
- 1950: Otto Diels / Kurt Alder
- 1951: Edwin McMillan / Glenn T. Seaborg
- 1952: Archer Martin / Richard Synge
- 1953: Hermann Staudinger
- 1954: Linus Pauling
- 1955: Vincent du Vigneaud
- 1956: Cyril Hinshelwood / Nikolay Semyonov
- 1957: Alexander Todd
- 1958: Frederick Sanger
- 1959: Jaroslav Heyrovský
- 1960: Willard Libby
- 1961: Melvin Calvin
- 1962: Max Perutz / John Kendrew
- 1963: Karl Ziegler / Giulio Natta
- 1964: Dorothy Hodgkin
- 1965: Robert Woodward
- 1966: Robert S. Mulliken
- 1967: Manfred Eigen / Ronald Norrish / George Porter
- 1968: Lars Onsager
- 1969: Derek Barton / Odd Hassel
- 1970: Luis Federico Leloir
- 1971: Gerhard Herzberg
- 1972: Christian B. Anfinsen / Stanford Moore / William Stein
- 1973: Ernst Otto Fischer / Geoffrey Wilkinson
- 1974: Paul Flory
- 1975: John Cornforth / Vladimir Prelog
- 1976: William Lipscomb
- 1977: Ilya Prigogine
- 1978: Peter D. Mitchell
- 1979: Herbert C. Brown / Georg Wittig
- 1980: Paul Berg / Walter Gilbert / Frederick Sanger
- 1981: Kenichi Fukui / Roald Hoffmann
- 1982: Aaron Klug
- 1983: Henry Taube
- 1984: Robert Merrifield
- 1985: Herbert A. Hauptman / Jerome Karle
- 1986: Dudley R. Herschbach / Yuan T. Lee / John Polanyi
- 1987: Donald J. Cram / Jean-Marie Lehn / Charles J. Pedersen
- 1988: Johann Deisenhofer / Robert Huber / Hartmut Michel
- 1989: Sidney Altman / Thomas Cech
- 1990: Elias Corey
- 1991: Richard R. Ernst
- 1992: Rudolph A. Marcus
- 1993: Kary Mullis / Michael Smith
- 1994: George Olah
- 1995: Paul J. Crutzen / Mario Molina / F. Sherwood Rowland
- 1996: Robert Curl / Harold Kroto / Richard Smalley
- 1997: Paul D. Boyer / John E. Walker / Jens Christian Skou
- 1998: Walter Kohn / John Pople
- 1999: Ahmed Zewail
- 2000: Alan J. Heeger / Alan MacDiarmid / Hideki Shirakawa
- 2001: William Knowles / Ryoji Noyori / K. Barry Sharpless
- 2002: John B. Fenn / Koichi Tanaka / Kurt Wüthrich
- 2003: Peter Agre / Roderick MacKinnon
- 2004: Aaron Ciechanover / Avram Hershko / Irwin Rose
- 2005: Robert H. Grubbs / Richard R. Schrock / Yves Chauvin
- 2006: Roger D. Kornberg
- 2007: Gerhard Ertl
- 2008: Osamu Shimomura / Martin Chalfie / Roger Y. Tsien
- 2009: Venkatraman Ramakrishnan / Thomas A. Steitz / Ada E. Yonath
- 2010: Richard F. Heck / Akira Suzuki / Ei-ichi Negishi
- 2011: Dan Shechtman
- 2012: Robert Lefkowitz / Brian Kobilka
- 2013: Martin Karplus / Michael Levitt / Arieh Warshel
- 2014: Eric Betzig / Stefan Hell / William E. Moerner
- 2015: Tomas Lindahl / Paul L. Modrich / Aziz Sancar
- 2016: Jean-Pierre Sauvage / Fraser Stoddart / Ben Feringa
- 2017: Jacques Dubochet / Joachim Frank / Richard Henderson
- 2018: Frances Arnold / Gregory Winter / George Smith
- 2019: John B. Goodenough / M. Stanley Whittingham / Akira Yoshino
- 2020: Emmanuelle Charpentier / Jennifer Doudna
- 2021: David MacMillan / Benjamin List
- 2022: Carolyn R. Bertozzi / Morten P. Meldal / Karl Barry Sharpless
- 2023: Moungi G. Bawendi / Louis E. Brus / Alexei I. Ekimov











