Protein-coding gene in the species Homo sapiens
| ATP5PF |
|---|
|
| Identifiers |
|---|
| Aliases | ATP5PF, ATP5, ATP5A, ATPM, CF6, F6, ATP synthase, H+ transporting, mitochondrial Fo complex subunit F6, ATP synthase peripheral stalk subunit F6, ATP5J |
|---|
| External IDs | OMIM: 603152; MGI: 107777; HomoloGene: 1272; GeneCards: ATP5PF; OMA:ATP5PF - orthologs |
|---|
| Gene location (Human) |
|---|
 | | Chr. | Chromosome 21 (human)[1] |
|---|
| | Band | 21q21.3 | Start | 25,716,503 bp[1] |
|---|
| End | 25,735,673 bp[1] |
|---|
|
| Gene location (Mouse) |
|---|
 | | Chr. | Chromosome 16 (mouse)[2] |
|---|
| | Band | 16|16 C3.3 | Start | 84,624,754 bp[2] |
|---|
| End | 84,632,513 bp[2] |
|---|
|
| RNA expression pattern |
|---|
| Bgee | | Human | Mouse (ortholog) |
|---|
| Top expressed in | - myocardium of left ventricle
- right ventricle
- Epithelium of choroid plexus
- cardiac muscle tissue of right atrium
- biceps brachii
- Skeletal muscle tissue of rectus abdominis
- body of tongue
- vena cava
- Skeletal muscle tissue of biceps brachii
- vastus lateralis muscle
|
| | Top expressed in | - right kidney
- digastric muscle
- saccule
- myocardium of ventricle
- thoracic diaphragm
- atrioventricular valve
- cardiac muscles
- facial motor nucleus
- triceps brachii muscle
- muscle of thigh
|
| | More reference expression data |
|
|---|
| BioGPS |  | | More reference expression data |
|
|---|
|
| Gene ontology |
|---|
| Molecular function | - transporter activity
- proton transmembrane transporter activity
- transmembrane transporter activity
- ATPase activity
- protein-containing complex binding
| | Cellular component | - mitochondrial proton-transporting ATP synthase complex, coupling factor F(o)
- proton-transporting ATP synthase complex, coupling factor F(o)
- membrane
- mitochondrial proton-transporting ATP synthase complex
- mitochondrial inner membrane
- mitochondrion
- extracellular space
- cell surface
| | Biological process | - ATP synthesis coupled proton transport
- ion transport
- substantia nigra development
- ATP metabolic process
- mitochondrial ATP synthesis coupled proton transport
- ATP biosynthetic process
- cristae formation
- transport
- positive regulation of heart rate
- response to muscle activity
- negative regulation of prostaglandin secretion
- positive regulation of blood pressure
- negative regulation of arachidonic acid secretion
| | Sources:Amigo / QuickGO |
|
| Orthologs |
|---|
| Species | Human | Mouse |
|---|
| Entrez | | |
|---|
| Ensembl | | |
|---|
| UniProt | | |
|---|
| RefSeq (mRNA) | NM_001003696
NM_001003697
NM_001003701
NM_001003703
NM_001685
|
|---|
NM_001320266
NM_001320267 |
| NM_016755
NM_001302213
NM_001302214
NM_001302215
NM_001302216
|
|---|
NM_001358499
NM_001358500 |
|
|---|
| RefSeq (protein) | NP_001003696
NP_001003697
NP_001003701
NP_001003703
NP_001307195
|
|---|
NP_001307196
NP_001676 |
| NP_001289142
NP_001289143
NP_001289144
NP_001289145
NP_058035
|
|---|
NP_001345428
NP_001345429 |
|
|---|
| Location (UCSC) | Chr 21: 25.72 – 25.74 Mb | Chr 16: 84.62 – 84.63 Mb |
|---|
| PubMed search | [3] | [4] |
|---|
|
| Wikidata |
| View/Edit Human | View/Edit Mouse |
|
| Mitochondrial ATP synthase coupling factor 6 (F6) |
|---|
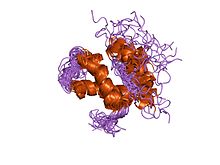 solution structure of subunit f6 from the peripheral stalk region of atp synthase from bovine heart mitochondria |
| Identifiers |
|---|
| Symbol | ATP-synt_F6 |
|---|
| Pfam | PF05511 |
|---|
| InterPro | IPR008387 |
|---|
| Available protein structures: |
|---|
| Pfam | structures / ECOD |
|---|
| PDB | RCSB PDB; PDBe; PDBj |
|---|
| PDBsum | structure summary |
|---|
|
ATP synthase-coupling factor 6, mitochondrial is an enzyme subunit that in humans is encoded by the ATP5PF gene.[5][6][7]
Function
Mitochondrial ATP synthase catalyzes ATP synthesis, utilizing an electrochemical gradient of protons across the inner membrane during oxidative phosphorylation. It is composed of two linked multi-subunit complexes: the soluble catalytic core, F1, and the membrane-spanning component, FO, which comprises the proton channel. The F1 complex consists of 5 different subunits (alpha, beta, gamma, delta, and epsilon) assembled in a ratio of 3 alpha, 3 beta, and a single representative of the other 3. The FO seems to have nine subunits (a, b, c, d, e, f, g, F6 and 8). This gene encodes the F6 subunit of the FO complex, required for F1 and FO interactions. Alternatively spliced transcript variants encoding different isoforms have been identified for this gene.[7]
The F6 subunit is part of the peripheral stalk that links the F1 and FO complexes together, and which acts as a stator to prevent certain subunits from rotating with the central rotary element. The peripheral stalk differs in subunit composition between mitochondrial, chloroplast and bacterial F-ATPases. In mitochondria, the peripheral stalk is composed of one copy each of subunits OSCP (oligomycin sensitivity conferral protein), F6, b and d.[8] There is no homologue of subunit F6 in bacterial or chloroplast F-ATPase, whose peripheral stalks are composed of one copy of the delta subunit (homologous to OSCP), and two copies of subunit b in bacteria, or one copy each of subunits b and b' in chloroplasts and photosynthetic bacteria.
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000154723 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000022890 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Higuti T, Tsurumi C, Kawamura Y, Tsujita H, Osaka F, Yoshihara Y, Tani I, Tanaka K, Ichihara A (Jul 1991). "Molecular cloning of cDNA for the import precursor of human coupling factor 6 of H(+)-ATP synthase in mitochondria". Biochemical and Biophysical Research Communications. 178 (2): 793–9. doi:10.1016/0006-291X(91)90178-A. PMID 1830479.
- ^ Javed AA, Ogata K, Sanadi DR (Jan 1991). "Human mitochondrial ATP synthase: cloning cDNA for the nuclear-encoded precursor of coupling factor 6". Gene. 97 (2): 307–10. doi:10.1016/0378-1119(91)90068-M. PMID 1825642.
- ^ a b "Entrez Gene: ATP5PF ATP synthase peripheral stalk subunit F6".
- ^ Carbajo RJ, Kellas FA, Runswick MJ, Montgomery MG, Walker JE, Neuhaus D (Aug 2005). "Structure of the F1-binding domain of the stator of bovine F1Fo-ATPase and how it binds an alpha-subunit". Journal of Molecular Biology. 351 (4): 824–38. doi:10.1016/j.jmb.2005.06.012. PMID 16045926.
External links
Further reading
- Kinosita K, Yasuda R, Noji H (2003). "F1-ATPase: a highly efficient rotary ATP machine". Essays in Biochemistry. 35: 3–18. doi:10.1042/bse0350003. PMID 12471886.
- Oster G, Wang H (Mar 2003). "Rotary protein motors". Trends in Cell Biology. 13 (3): 114–21. doi:10.1016/S0962-8924(03)00004-7. PMID 12628343.
- Leyva JA, Bianchet MA, Amzel LM (2003). "Understanding ATP synthesis: structure and mechanism of the F1-ATPase (Review)". Molecular Membrane Biology. 20 (1): 27–33. doi:10.1080/0968768031000066532. PMID 12745923. S2CID 218895820.
- Hochstrasser DF, Frutiger S, Paquet N, Bairoch A, Ravier F, Pasquali C, Sanchez JC, Tissot JD, Bjellqvist B, Vargas R (Dec 1992). "Human liver protein map: a reference database established by microsequencing and gel comparison". Electrophoresis. 13 (12): 992–1001. doi:10.1002/elps.11501301201. PMID 1286669. S2CID 23518983.
- Yan WL, Lerner TJ, Haines JL, Gusella JF (Nov 1994). "Sequence analysis and mapping of a novel human mitochondrial ATP synthase subunit 9 cDNA (ATP5G3)". Genomics. 24 (2): 375–7. doi:10.1006/geno.1994.1631. PMID 7698763.
- Elston T, Wang H, Oster G (Jan 1998). "Energy transduction in ATP synthase". Nature. 391 (6666): 510–3. Bibcode:1998Natur.391..510E. doi:10.1038/35185. PMID 9461222. S2CID 4406161.
- Webster KA, Oliver NA, Wallace DC (Mar 1982). "Assignment of an oligomycin-resistance locus to human chromosome 10". Somatic Cell Genetics. 8 (2): 223–44. doi:10.1007/BF01538679. PMID 9732751. S2CID 12445064.
- Wang H, Oster G (Nov 1998). "Energy transduction in the F1 motor of ATP synthase". Nature. 396 (6708): 279–82. Bibcode:1998Natur.396..279W. doi:10.1038/24409. PMID 9834036. S2CID 4424498.
- Wiemann S, Weil B, Wellenreuther R, Gassenhuber J, Glassl S, Ansorge W, Böcher M, Blöcker H, Bauersachs S, Blum H, Lauber J, Düsterhöft A, Beyer A, Köhrer K, Strack N, Mewes HW, Ottenwälder B, Obermaier B, Tampe J, Heubner D, Wambutt R, Korn B, Klein M, Poustka A (Mar 2001). "Toward a catalog of human genes and proteins: sequencing and analysis of 500 novel complete protein coding human cDNAs". Genome Research. 11 (3): 422–35. doi:10.1101/gr.GR1547R. PMC 311072. PMID 11230166.
- Osanai T, Nakamura M, Sasaki S, Tomita H, Saitoh M, Osawa H, Yamabe H, Murakami S, Magota K, Okumura K (Dec 2003). "Plasma concentration of coupling factor 6 and cardiovascular events in patients with end-stage renal disease". Kidney International. 64 (6): 2291–7. doi:10.1046/j.1523-1755.2003.00334.x. PMID 14633154.
- Cross RL (Jan 2004). "Molecular motors: turning the ATP motor". Nature. 427 (6973): 407–8. Bibcode:2004Natur.427..407C. doi:10.1038/427407b. PMID 14749816. S2CID 52819856.
- Jacobs LJ, de Coo IF, Nijland JG, Galjaard RJ, Los FJ, Schoonderwoerd K, Niermeijer MF, Geraedts JP, Scholte HR, Smeets HJ (Mar 2005). "Transmission and prenatal diagnosis of the T9176C mitochondrial DNA mutation". Molecular Human Reproduction. 11 (3): 223–8. CiteSeerX 10.1.1.603.8165. doi:10.1093/molehr/gah152. PMID 15709156.
This article incorporates text from the public domain
Pfam and
InterPro: IPR008387

















